Оперативное планирование доставки вакцин
Ларин О.Н.1,2 ![]() , Матосов М.В.1
, Матосов М.В.1 ![]()
1 Российский экономический университет им. Г.В. Плеханова, Москва, Россия
2 Российский университет транспорта (МИИТ), Москва, Россия
Скачать PDF | Загрузок: 7
Статья в журнале
Экономика, предпринимательство и право (РИНЦ, ВАК)
опубликовать статью | оформить подписку
Том 16, Номер 3 (Март 2026)
Эта статья проиндексирована РИНЦ, см. https://elibrary.ru/item.asp?id=89188438
Аннотация:
Участники цепочек поставок вакцин планируют взаимодействие, как правило, на основе долгосрочных договоров, предусматривающих поставки лекарственных препаратов с установленной периодичность в заданных объемах с использованием одного или нескольких видов транспорта. Однако в чрезвычайных ситуациях поставщики вакцин вынуждены корректировать действующие планы с учётом потребностей новых получателей. Поэтому логистические компании заинтересованы в использовании передовых решений, позволяющих определять оптимальные варианты доставки вакцин всем получателем в полном объеме в установленные сроки в условиях ограниченного количества логистических ресурсов. Для оперативного планирования доставки вакцин предлагается использовать разработанную математическую модель, которая определяет оптимальные по стоимости варианты доставки от поставщика получателям, а также позволяет определять потребное количество логистических ресурсов, необходимое для полного выполнения заявленных объемов. Разработанная модель также может использоваться в качестве основы для разработки моделей комплексного планирования доставки вакцин от множества поставщиков различным получателям
Ключевые слова: холодовая цепь, способ доставки, логистика вакцин, оперативное планирование
JEL-классификация: L81, M11, D24
Введение
В современных условиях (рост миграции населения и объемов международных сообщений) распространение инфекционных болезней приобретает глобальный характер. Своевременная иммунопрофилактика позволяет существенно снизить заболеваемость населения опасными инфекционными болезнями и ограничить распространение инфекций. Такой эффект вакцинопрофилактики достигается при значительном охвате (более 95 процентов) профилактическими прививками населения [2].
Проведение вакцинации населения организуется, как правило, в плановом порядке в рамках национального календаря прививок и календаря профилактических прививок по эпидемическим показаниям. Однако в современных условиях многие новые инфекционные заболевания стремительно распространяются по регионам и разным странам. Поэтому к работе логистических компаний, которые обслуживают холодовые цепи поставок иммунобиологических лекарственных препаратов (далее – холодовая цепь вакцин, ХЦВ), предъявляются высокие требования по обеспечению своевременности доставки вакцин при безусловной сохранности груза и низкой стоимости услуг.
В последние годы отмечается рост объемов поставок вакцин. При этом структура закупок вакцин варьируется из года в год в зависимости от многих факторов: эпидемическая ситуация, уровень рождаемости и т.д. Кроме того изменяется источники поставок. Например, отечественная вакцина для профилактики гриппа «Совигрипп», которую производит «Нацимбио», в 2024 году входила в тройку лидеров по стоимостному объему в сегменте закупок для нужд лечено-профилактических учреждений и аптечных сетей. Однако в сравнении с 2023 г. этот показатель снизился на 14%, так как произошло перераспределение закупок на противогриппозные аналоги «Ультрикс» и «Флю-М», продажи которых для больниц увеличились на 49,7 и 1263,2 процента соответственно [8]. Изменение поставщиков также требует формирования новых эффективных логистических цепочек с учетом особенностей наличной инфраструктуры для проектирования маршрутов перевозок, эксплуатационных характеристик располагаемого подвижного состава, возможных сезонных ограничений на перевозки и других факторов.
Комплекс задач по развитию ХЦВ и процессов оказания логистических услуг на всех этапах данной цепочки предусмотрен рядом руководящих документов. Согласно Стратегии развития фармацевтической промышленности Российской Федерации на период до 2030 года предусмотрено развитие производства лекарственных средств на территории страны как для внутреннего рынка, так и для реализации экспортного потенциала фармацевтической промышленности [1]. В связи с этим одним из ключевых направлений данной Стратегии является выстраивание стабильных холодовых цепочек, способных обеспечить физическую и экономическую доступность лекарственных препаратов, в том числе вакцин, на отечественном и зарубежном рынках.
Основными особенностями ХЦВ являются: необходимость поддержания строгих температурных режимов для обеспечения сохранности вакцин; высокие затраты на логистику; жесткие требования к сроку доставки вакцин, чтобы обеспечить их сохранность и своевременность оказания помощи населению; различия потребностей в местах вакцинации, которые могут располагаться на значительном удалении друг от друга [17] (Lotty et al., 2018).
Во многих исследованиях отмечается, что изменение плановых параметров поставок вакцин создает дополнительные требования к ХЦВ и приводит к дефициту логистических ресурсов на отдельных звеньях цепочки (см. [11, 25] (De et al., 2008; Terri et al., 2012) и др.). Как следствие, ХЦВ не способны обеспечивать бесперебойные и эффективные поставки вакцин в объеме, соответствующем требуемому уровню оказания медицинской помощи [21, 29] (Patrick et al., 2015). В рамках данного исследования под необходимыми логистическими ресурсами понимается, прежде всего, достаточное количество подвижного состава разных видов транспорта, который используется для своевременного и сохранного перемещения вакцин между звеньями ХЦВ.
При дефиците логистических ресурсов налаженные ХЦВ начинают работать со сбоями в время чрезвычайных ситуаций, в результате чего существенно возрастают расходы на логистику, что ставит под угрозу современность реализации мер по защите населения от инфекций [14, 15, 19, 23, 26] (Justin et al., 2021; Kartoglu et al., 2020; Nader et al., 2024; Stef et al., 2016; Tina-Marie et al., 2012). Под ограниченностью наличных логистических ресурсов понимается нехватка транспортных средств, плохое состояния инфраструктуры и пр. Авторы [9] (Ashvin et al., 2017), на основе изучения опыта реализации национальных программ иммунизации в ряде стран Азии и Африки, сделали вывод, что существующие системы ХЦВ не способны гарантировать наличие вакцин из-за недостаточной и неоптимальной пропускная способности звеньев ХЦВ. В исследовании [12] (Thomas et al., 2021) отмечается, что во многих странах ХЦВ не обладают достаточными логистическими ресурсами для достижения текущих целевых показателей вакцинации.
Планирование поставок вакцин предполагает систематическое принятие ключевых решений как на стратегическом, так и на тактическом уровнях Авторы [17] разделяют задачи планирования в ХЦВ на четыре группы: продукт (какую вакцину следует поставлять), производство (откуда следует поставлять), назначение (сколько и в какие сроки следует поставлять) и распределение (как следует перевозить). В рамках данного исследования рассматриваются вопросы четвертой группы, а именно оперативного планирования доставки вакцин в условиях ограниченных логистических ресурсов.
Ввиду временного характера чрезвычайных ситуаций капитальная модернизация инфраструктуры отдаленных регионов нецелесообразна. Поэтому актуальной задачей является разработка методов оперативного планирования наименее затратных вариантов доставки вакцин с использованием разных видов транспорта в условиях ограниченного количества специализированного подвижного состава.
Вопросы выбора оптимальных вариантов доставки вакцин относятся к числу приоритетных у многих исследователей [20, 22, 24, 32] (Nugroho et al., 2022; Rafael et al., 2026; Tan et al., 2014; Shi et al., 2022). При планировании широко используются математические модели выбора оптимальных вариантов доставки по одному или нескольким критериям. Найденное решение должно обеспечивать низкие операционные затраты и одновременно быструю доставку вакцин конечным потребителям, особенно во время пандемии.
Особенности применения математических методов для оптимального планирования доставки вакцин в рамках плановой программы иммунизации рассмотрены в работе [26] (Tina-Marie et al., 2012). Авторы использовали вычислительную дискретно-событийную имитационную модель цепочки поставок. Их решение позволяет выявить узкие места в цепочке поставок, которые снижают доступность вакцин. Для решения данной проблемы авторы изменяют длительность периодов поставок.
Имитационное моделирование также может применяться для оценки ключевых показателей ХЦВ, таких как уровень обслуживания, экономическая эффективность и экологичность. Результаты имитационного моделирования показывают прямую зависимость данных показателей от размера парка транспортных средств, используемых для доставки вакцин, типа подвижного состава и маршрутов перевозок [26] (Tina-Marie et al., 2012). Соответственно, данные факторы будем учитывать при планировании доставки вакцин.
На операционном уровне задача выбора способа доставки является одной из важнейших проблем, влияющих на эффективность распределения вакцин с минимальными затратами. Наиболее часто в задачах планирования доставки товаров в холодовой цепи применяется различные методы маршрутизации транспортных средств, которые используют различные ограничения, например, временные окна отправки для каждого клиента, применение нескольких типов подвижного состава с отличающимися показателями энергопотребления, вместимости и т.п. Применение алгоритмов маршрутизации транспортных средств повышает своевременность доставки вакцин более чем на 10% [19] (Nader et al., 2024).
Выполненный анализ приведенных выше и многих других работ показал, что для оперативного реагирования на изменение плановых направлений и объемов доставки логистические операторы используют различные транспортно-технологические схемы доставки вакцин между звеньями ХЦВ, в том числе с использованием разных видов транспорта. Однако вопросы выбора наименее затратных вариантов доставки вакцин одним и несколькими видами транспорта в установленные сроки в условиях ограниченного количества логистических ресурсов остаются недостаточно исследованными.
Целью исследования является разработка математической модели формирования логистических параметров оперативного плана поставок вакцин из организаций оптовой торговли федерального масштаба на региональные распределительные склады. Основными методами исследования являются сравнительный анализ, системный подход, моделирование и целочисленное программирование.
Модель цепи поставок вакцин
Структурные и технологические особенности цепей поставок, а также подходы к организации взаимодействия их участников, рассмотрены в работах многих отечественных и зарубежных ученых (см. например, [5–7, 10, 13, 16, 18, 27, 31] (Matosov et al., 2023; Azoev et al., 2020; Abramova et al., 2024; Bruce et al., 2017; Haidari et al., 2013; Lim et al., 2022; Mercy et al., 2017; Ulla et al., 2009; Yauba et al., 2020) и др.). По мнению экспертов Всемирной организации здравоохранения, ключевыми компонентами ХЦВ являются:
• персонал, который должен быть обучен для управления хранением и распределением вакцин на всех этапах цепочки;
• специализированное оборудование, которое применяется для хранения и транспортирования вакцин и контроля соблюдения температурного режима;
• технологии, использование которых должно обеспечивать безопасную и эффективную эксплуатацию оборудования и выполнение процессов хранения и транспортировки вакцин [28].
Структура ХЦВ отображается в виде многоуровневой сетевой модели [4] (Larin et al., 2024). Как правило, используются трех и четырех уровневые структуры ХЦВ. В состав трехуровневой модели входят участники ХЦВ на национальном, региональном и местном уровнях [31] (Yauba et al., 2020). Плановое распределение вакцин с национального на региональные уровни (оптовые склады) осуществляется по принципу «выталкивания», а с регионального уровня вакцины доставляются на местный уровень (в лечено-профилактические учреждения) по запросу.
В ряде стран, в том числе и в России, используется четырехуровневая структура ХЦВ в составе национального, регионального, районного и клинического уровней [3]:
1) первый уровень – от отечественных производителей или импортеров вакцины доставляются до организаций оптовой торговли федерального масштаба;
2) второй уровень – хранение вакцин на складах оптовых поставщиков и доставка до крупных региональных дистрибьюторов;
3) третий уровень – хранение вакцин на складах региональных дистрибьюторов и доставка вакцин до лечено-профилактических учреждений или их обособленных подразделений, использующих или реализующих вакцины в розницу;
4) четвертый уровень – хранение вакцин в лечено-профилактических учреждениях, где проводится иммунизация.
Схема «уровней» ХЦВ и направления поставок вакцин показаны на рисунке.
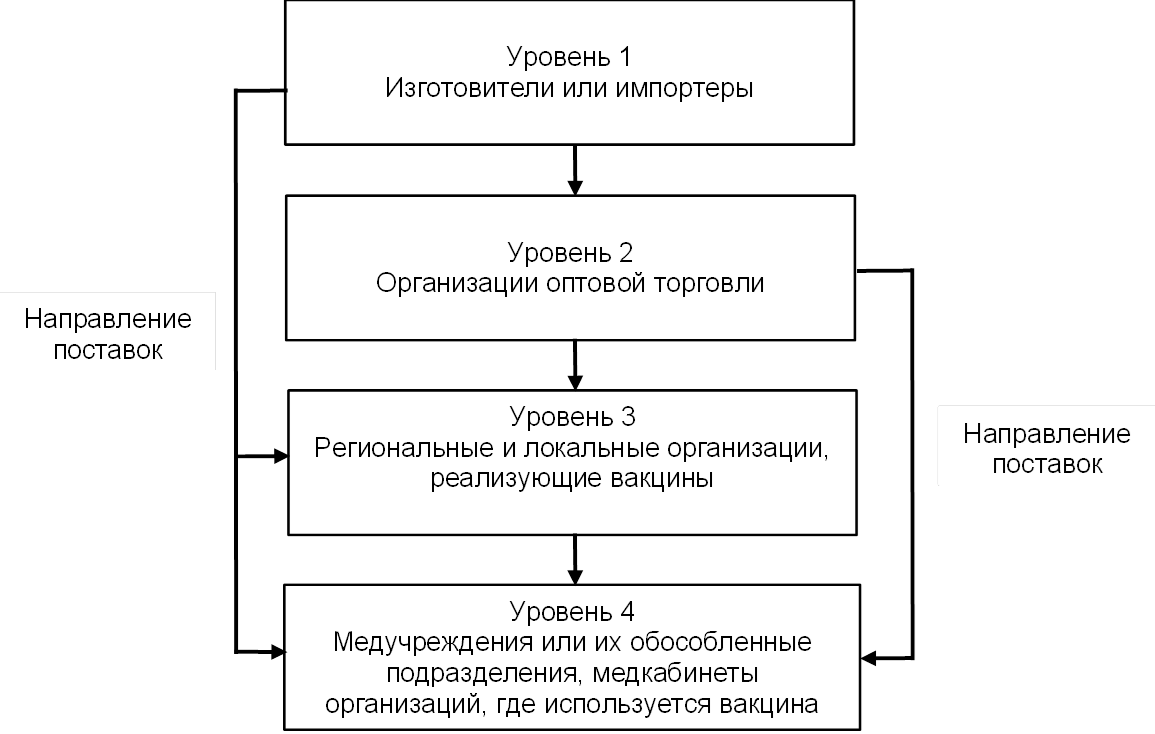
Рисунок. Схема «уровней» ХЦВ
Источник: составлено авторами с учетом требований [3].
Поставка вакцин между первым и вторым уровнями ХЦВ осуществляется крупными партиями, как правило, одним способом доставки на значительные расстояния. Поставка с третьего на четвертый уровень ХЦВ осуществляется, как правило, на небольшие расстояния мелкими партиями с использованием автомобильного транспорта. Наибольшая вариативность плановых параметров и используемых вариантов доставки присуща поставкам со второго на третий уровень ХЦВ. Выполненный выше обзор научных работ показывает, что поставки между вторым и третьим уровнями характеризуются частыми сбоями, высокими затратами и дефицитом логистических ресурсов. Поэтому будет разработана математическая модель выбора оптимального оперативного плана доставки вакцин со второго на третий уровень ХЦВ.
Математическая модель оперативного планирования
Основным компонентом модели является «способ доставки», под которым понимается организационно-технологическая система, использующая комплекс технических и инфраструктурных объектов, обеспечивающих физическое перемещения грузов от отправителя до получателя. В рамках данного исследования рассматривается доставка вакцин от склада единственного поставщика (дистрибьютор на 2 уровне ХЦВ) на склад единственного получателя (лечено-профилактическое учреждение на 3 уровне ХЦВ).
Выделение различных способов доставки основано на вариативности использования видов транспорта. Два базовых типа способов доставки образуются в зависимости от числа видов транспорта, которыми осуществляется транспортирование вакцин. Первый «прямой» способ предусматривает доставку только одним доступным видом транспорта «от двери до двери». В рамках прямого способа могут выделятся отдельные разновидности доставки, например, только автомобильным, либо только воздушным (дроном) транспортом.
Второй «смешанный» тип способа доставки вакцин от отправителя до получателя основан на использовании нескольких видов транспорта (смешанный многомодальный) или одного вида транспорта с участием различных типов подвижного состава (смешанный одномодальный). При смешанном многомодальном способе (синоним мультимодальной перевозки) доставка представляется собой комбинацию разных видов транспорта и типов транспортных средств. Например, на «первой миле» груз доставляется со склада малотоннажным автомобилем на грузовой терминал, где контейнеры перегружаются в самолет (или морское судно), которое осуществляет перевозку вакцин в терминал назначения в отдаленном регионе. На «последней миле» груз снова доставляется автомобилем от терминала до склада получателя. При смешанном одномодальном типе доставка осуществляется различными малотоннажными и крупнотоннажными изотермическими автофургонами с рефрижераторной установкой, которые имеют грузоподъемность от 0,5 и более тонн. На короткие расстояния, в пределах города, вакцины развозятся получателям автомобилями грузоподъемность до одной тонны. При необходимости, один автомобиль в течение суток может выполнить несколько рейсов и доставить груз в адрес нескольких получателей. Для доставки на большие расстояния в отдаленные регионы используются транспортные средства с грузоподъемностью свыше 5 тонн.
Для целей планирования и моделирования доставки вакцин с различными способами будем использовать следующие показатели:
– фактический срок доставки вакцин от склада отправителя до склада получателя k-м способом (k Î K, K = 1, 2, … n)– Тф(k);
– плановый срок доставки вакцин от склада отправителя до склада получателя – Тр;
– плановый объем поставок вакцин от склада отправителя до склада получателя Qр(k);
– базовый объем доставки вакцин от склада отправителя до склада получателя k-м способом Qб(k);
– фактический объем доставки вакцин от склада отправителя до склада получателя k-м способом Qф(k);
– общий фактический объем доставки вакцин от склада отправителя до склада получателя всеми k-ми способами Qф;
Фактическая продолжительность доставки Tф(k) учитывает все затраты времени на начальные и конечные операции (погрузка, выгрузка, оформление документов и пр.), а также время движения транспортных средств по маршруту (при прямом способе) или по отдельным участкам многомодального маршрутам (при смешанном способе).
При многомодальной перевозке фактическая продолжительность доставки Tф(k) определяется как сумма фактических продолжительностей доставки Tфw(k) на отдельных участках многомодального маршрута, обслуживаемых разными w-ми (w Î W, W = 1, 2, … m) видами транспорта:
![]() . (1)
. (1)
Плановая продолжительность доставки Tр устанавливается в договоре на доставку и должна соблюдаться логистическим оператором. Поэтому фактическая продолжительность любым способом Tф(k) не должна превышать плановую:
"k Î Y, Tф(k) ≤ Tр; Y Í K. (2)
Базовый объем доставки Qб(k) по маршруту k-ым способом характеризует единичный (минимально допустимый) объем отправки для соответствующего k-ого способа. Единичный объем отправок может измеряться в килограммах, тоннах, штуках, термоконтейнерах, кубометрах и т.п. Размерность определяется с учетом организационных и технологических особенностей доставки, используемой упаковки, установленного порядка учета вакцин и т.д.
Например, если доставка осуществляется сборными LTL-отправками (Less Than Truck Load), то базовый объем будет менее грузоподъемности (грузовместимости) qw(k) подвижного состава выбранного вида транспорта:
Qбw(k) < qw(k), (3)
а при полнозагруженных FTL–отправках (Full Truck Load) базовый объем будет равен грузоподъемности (грузовместимости) транспортного средства:
Qбw(k) = qw(k). (4)
Например, для перевозки вакцин применяются специальные термоконтейнеры емкостью от 1 до 100 литров. Соответственно, при LTL-отправке в автофургоне вместимостью 100 литров базовый объем может быть равен 1 и более литрам, а при FTL-отправке базовый объем составит 100 литров.
При многомодальной перевозке базовые объемы Qбw(k) поставок каждым w-м видом транспорта на участках многомодального маршрута отличаются, так как грузоподъемность (грузовместимость) подвижного состава qw(k) разных видов транспорта отличается. При этом базовые объемы Qбw(k) по w-м участкам каждого k-го способа должны быть равны, чтобы соблюдался баланс между перевезенными грузов по участкам: например, автомобили от склада поставщика завозят вакцины на многомодальный терминал, из которого все завезенные вакцины должны быть вывезены другим видом транспорта, например, воздушным. Для выполнения данного условия базовые объемы на всех участках многомодального маршрута должны быть уравнены по максимальному значению базового объема Qбw(k) на одном из участков:
![]() . (5)
. (5)
На основе базового объема Qб(k) определяются минимально необходимое количество подвижного состава на многомодальном aбw(k) маршруте, при которых обеспечивается указанное выше равенство базовых объемов на всех участках:
aбw(k) = Qб(k) / Qбw(k). (6)
На одномодальном маршруте базовый объем Qб(k) равен Qбw(k) и с учетом (6) минимально необходимое количество подвижного состава aбw(k) принимается равным единице.
Фактически объем доставки Qф(k) определяется на основе базового объема и равен его суммарному значению Qб(k) для всех отправок u(k), которые запланированы к перевозке соответствующим k-м способом за фактический срок Тф(k):
Qф(k) = Qб(k) ∙ u(k). (7)
Фактическое количество единиц каждого вида транспорта aфw(k) на одномодальном и многомодальном маршруте, которое используется для перевозки Qф(k), определяется по формуле:
aфw(k) = aбw(k) ∙ u(k). (8)
Для выполнения запланированных отправок u(k) число фактически необходимых транспортных средств афw(k) каждого вида транспорта не должно превышать количество наличного подвижного состава Аw(k):
афw(k) ≤ Аw(k). (9)
Величина Аw(k) используется в модели планирования поставок в качестве ограничения.
Общий фактический объем Qф доставки вакцин от склада отправителя до склада получателя всеми k-ми способами рассчитывается по формуле:
![]() . (10)
. (10)
и должен соответствовать плановому объему поставок Qр:
Qф = Qр. (11)
Критерием оптимального варианта доставки является величина операционной прибыли H, которая определяется как разница между стоимостью контракта Z, который заключил логистический оператор с поставщиком на доставку планового объема вакцин Qр, и общей величиной затрат G логистического оператора, которые он понесет на выполнение данной доставки. Запишем целевую функцию оптимизационной модели:
H = ( Z – G) → max. (12)
Затраты G рассчитываются через произведение фактического объема Qф(k) на норму затрат g(k) на единицу груза для соответствующих способов доставки:
![]() , руб., (13)
, руб., (13)
![]() , руб., (14)
, руб., (14)
где gw(k) – норма затрат для w-х видов транспорта на k-м способе, (руб./ед.):
В выражении (13) норма затрат g(k) имеет фиксированное значение, а фактические объемы Qф(k), согласно (7), изменяется в зависимости от количества назначенных отправок u(k). Поэтому переменной в функции H является количество отправок.
Так как стоимость контракта Z логистического оператора является фиксированной величиной, то решение задачи (12) сводится к поиску оптимального плана доставки F*:
F* = { Qф(k); u(k) }→ opt, (15)
который представляет собой вектор оптимальных параметров Qф*(k) и u*(k), при которых затраты достигают минимального значения и, как следствие, максимальной операционной прибыли:
G = { F* }→ min. (16)
Запишем математическую модель оптимального плана доставки вакцин от поставщика получателям:
![]() , (17)
, (17)
при aфw(k) ≤ Аw(k); Qф = Qр; Tф(k) ≤ Tр;
где uy – целое; y Î Y (Y = 1,N); k Î Y; Y Í K (K = 1, 2, … n); w Î W (W = 1, 2, … m).
Данная задача может быть решена известными методами целочисленного программирования. Причем у некоторых полученных решений при минимальных затратах могут не соблюдаться все ограничения, на что следует обратить внимание. Например, если не соблюдается ограничение по равенству планового и фактического объемов доставки, это значит, что логистический оператор не сможет выполнить условия контракта в полном объеме. Такая ситуация образуется при недостаточном количестве подвижного состава Аw(k). Для определения количества транспортных средств Аw*(k), необходимых для полного выполнения контрактных обязательств (11), следует задачу (17) решать без ограничения по Аw(k). Разница между Аw*(k) и Аw(k) покажет на сколько единиц транспортных средств логистический оператор должен увеличить свой парк.
Заключение
В статье представлен подход к разработке оптимальных по затратам оперативных планов поставок вакцин. Для перевозки максимально возможного объема вакцин при ограниченных логистических ресурсах грузы могут перераспределяться между несколькими способами доставки, чтобы обеспечить своевременное наличие лекарственных средств в очагах инфекций. Представленная математическая модель оперативного планирования поставок позволяет определять потребное количество логистических ресурсов для доставки всего необходимого объема вакцин. Данная модель также может использоваться в качестве основы для разработки моделей комплексного планирования доставки вакцин от множества поставщиков различным получателям в местах эпидемий.
Источники:
2. Стратегия развития иммунопрофилактики инфекционных болезней на период до 2035 года» – утв. Распоряжением Правительства РФ от 18.09.2020 № 2390-р.
3. СанПиН 3.3686-21 «Санитарно-эпидемиологические требования по профилактике инфекционных болезней» – утв. Постановлением Главного государственного санитарного врача РФ от 28.01.2021 № 4.
4. Ларин О. Н., Матосов М. В. Логистика вакцин: проблемы и перспективы цифровизации // Логистика. – 2024. – № 10. – c. 46-52.
5. Матосов М.В., Ларин О.Н. Организация взаимодействия участников фармацевтической цепочки поставок // Экономика, предпринимательство и право. – 2023. – № 4. – c. 983-994. – doi: 10.18334/epp.13.4.117492.
6. Азоев Г. Л., Быкова О.Н., Гарнов А.П. Развитие экономических систем в цифровой экономике: маркетинг, сфера услуг, логистика. - Москва : Общество с ограниченной ответственностью Русайнс, 2020. – 262 c.
7. Абрамова Е.Р., Асалиев А.М., Глинская М. И. Устойчивость цепей поставок в условиях цифровой трансформации бизнеса. - Москва : Общество с ограниченной ответственностью Русайнс, 2024. – 304 c.
8. Фармацевтический рынок России 2024. DSM Group, 2025. – 132 с. [Электронный ресурс]. URL: https://dsm.ru/upload/iblock/70c/jge20mm780rph1l9p1d6ucvg95kkxdtf.pdf (дата обращения: 01.01.2026).
9. Ashvin Ashok, Michael Brison, Yann LeTallec Improving cold chain systems: Challenges and solutions // Vaccine. – 2017. – № 17. – p. 2217-2223. – doi: 10.1016/j.vaccine.2016.08.045.
10. Bruce Y. Lee, Leila A. Haidari The importance of vaccine supply chains to everyone in the vaccine world // Vaccine. – 2017. – № 35. – p. 4475-4479. – doi: 10.1016/j.vaccine.2017.05.096.
11. De Oliveira L. H., Danovaro-Holliday M. C., Matus C. R., Andrus J. K. Rotavirus vaccine introduction in the Americas: progress and lessons learned // Expert Review of Vaccines. – 2008. – № 7. – p. 345-353. – doi: 10.1586/14760584.7.3.345.
12. Thomas D., Motomoke E., Crawford J., Defawe O., Makaya A., Ngwato J.W., Bompongo J., Monzembela J., Ailstock G., Bancroft E., Magadzire B., Baabo D., Watson N. Optimized supply chain model reduces health system costs in DRC // Vaccine. – 2021. – № 30. – p. 4166-4172. – doi: 10.1016/j.vaccine.2021.05.083.
13. Haidari L.A., Connor D.L., Wateska A.R., Brown S.T., Mueller L.E. Augmenting Transport versus Increasing Cold Storage to Improve Vaccine Supply Chains // PLOS ONE. – 2013. – № 5. – p. e64303. – doi: 10.1371/journal.pone.0064303.
14. Justin R. Ortiz, Joanie Robertson, Jui-Shan Hsu, Stephen L. Yu, Amanda J. Driscoll, Sarah R. Williams, Wilbur H. Chen, Meagan C. Fitzpatrick, Samba Sow, Robin J. Biellik, Kathleen M. Neuzil The potential effects of deploying SARS-Cov-2 vaccines on cold storage capacity and immunization workload in countries of the WHO African Region // Vaccine. – 2021. – № 15. – p. 2165-2176. – doi: 10.1016/j.vaccine.2021.02.037.
15. Kartoglu U.H., Moore K.L., Lloyd J.S. Logistical challenges for potential SARS-CoV-2 vaccine and a call to research institutions, developers and manufacturers // Vaccine. – 2020. – № 34. – p. 5393-5395. – doi: 10.1016/j.vaccine.2020.06.056.
16. Lim J., Norman B.A., Rajgopal J. Redesign of vaccine distribution networks // Intl. Trans. in Op. Res.. – 2022. – № 29. – p. 200-225. – doi: 10.1111/itor.12758.
17. Lotty Evertje Duijzer, Willem van Jaarsveld, Rommert Dekker Literature review: The vaccine supply chain // European Journal of Operational Research. – 2018. – № 1. – p. 174-192. – doi: 10.1016/j.ejor.2018.01.015.
18. Mercy Mvundura // The Pan African Medical Journal. – 2017. – № 3. – p. 27. – url: https://www.panafrican-med-journal.com/content/series/27/3/27/pdf/27.pdf.
19. Nader Al Theeb, Mohammed Abu-Aleqa, Ali Diabat Multi-objective optimization of two-echelon vehicle routing problem: Vaccines distribution as a case study // Computers & Industrial Engineering. – 2024. – p. 109590. – doi: 10.1016/j.cie.2023.109590.
20. Nugroho Agung Pambudi, Alfan Sarifudin, Indra Mamad Gandidi, Rahmat Romadhon Vaccine cold chain management and cold storage technology to address the challenges of vaccination programs // Energy Reports. – 2022. – p. 955-972. – doi: 10.1016/j.egyr.2021.12.039.
21. Patrick Lydon, Ticky Raubenheimer, Michelle Arnot-Krüger, Michel Zaffran Outsourcing vaccine logistics to the private sector: The evidence and lessons learned from the Western Cape Province in South-Africa // Vaccine. – 2015. – № 29. – p. 3429-3434. – doi: 10.1016/j.vaccine.2015.03.042.
22. Rafael Arevalo-Ascanio, Elena Prozorova, Annelies De Meyer, Roel Gevaers, Ruben Guisson, Wouter Dewulf Environmental and social assessment of transport and cooling systems in last-mile cold-chain vaccine distribution: A case study in Flanders, Belgium // Research in Transportation Economics. – 2026. – p. 101693. – doi: 10.1016/j.retrec.2025.101693.
23. Stef Lemmens, Catherine Decouttere, Nico Vandaele, Mauro Bernuzzi A review of integrated supply chain network design models: Key issues for vaccine supply chains // Chemical Engineering Research and Design. – 2016. – p. 366-384. – doi: 10.1016/j.cherd.2016.02.015.
24. Tan L.J., Barnett M.A., Eisenberg A., Fox Fields H., Hackell J.M., Léger M.M., Meadows B., Moore K.L., Rehm S.J., Rothholz M.C., Smith C., Talkington K., Wexler D.L. From refrigerator to arm: Issues in vaccination delivery // Vaccine. – 2014. – № 21. – p. 2389-2393. – doi: 10.1016/j.vaccine.2014.02.045.
25. Terri B. Hyde, Holly Dentz, Susan A. Wang, Helen E. Burchett, Sandra Mounier-Jack, Carsten F. Mantel The impact of new vaccine introduction on immunization and health systems: A review of the published literature // Vaccine. – 2012. – № 45. – p. 6347-6358. – doi: 10.1016/j.vaccine.2012.08.029.
26. Tina-Marie Assi, Korngamon Rookkapan, Jayant Rajgopal, Vorasith Sornsrivichai, Shawn T. Brown, Joel S. Welling How influenza vaccination policy may affect vaccine logistics // Vaccine. – 2012. – № 30. – p. 4517-4523. – doi: 10.1016/j.vaccine.2012.04.041.
27. Ulla K. Griffiths, Viola S. Korczak, Dereje Ayalew, Asnakew Yigzaw Incremental system costs of introducing combined DTwP–hepatitis B–Hib vaccine into national immunization services in Ethiopia // Vaccine. – 2009. – № 9. – p. 1426-1432. – doi: 10.1016/j.vaccine.2008.12.037.
28. Unit 4 : Cold chain and logistics management: Immunization handbook for Medical Officers. [Электронный ресурс]. URL: https://cdn.who.int/media/docs/default-source/searo/india/publications/immunization-handbook-107-198-part2.pdf (дата обращения: 21.01.2026).
29. World Health Organization et al. Effective vaccine management (EVM) initiative //EVM-setting standards for the Immunization Supply Chain. – 2011. – Т. 14.
30. Xu Sun, Eugenia Ama Andoh, Hao Yu. A simulation-based analysis for effective distribution of COVID-19 vaccines: A case study in Norway // Transportation Research Interdisciplinary Perspectives. – 2021. – p. 100453. – doi: 10.1016/j.trip.2021.100453.
31. Yauba S, Joelle S, Jude N, Tracy BO, Marie K, et al. (2018) Temperature Monitoring in the Vaccine Cold Chain in Cameroon. J Vaccines Vaccin 9: 384. doi:10.4172/2157-7560.1000384, World Health Organization et al. Vaccine management and logistics support //World Health Organization (WHO). – 2020. – С. 1-15
32. Shi Yu., Lin Yu., Lim M.K., Tseng M.L., Tan Ch., Li Ya. An intelligent green scheduling system for sustainable cold chain logistics // Expert Systems with Applications. – 2022. – p. 118378. – doi: 10.1016/j.eswa.2022.118378.
Страница обновлена: 19.05.2026 в 06:33:50
Download PDF | Downloads: 7
Operational planning of vaccine delivery
Larin O.N., Matosov M.V.Journal paper
Journal of Economics, Entrepreneurship and Law
Volume 16, Number 3 (March 2026)
Abstract:
Participants in vaccine supply chains typically plan their interactions based on long-term contracts providing for the delivery of drugs at set intervals in specified volumes using one or more modes of transport. However, in emergency situations, vaccine suppliers are forced to adjust existing plans to accommodate the needs of new recipients. Therefore, logistics companies are interested in using advanced solutions that enable them to determine optimal options for delivering vaccines to all recipients in full, within the specified timeframe, given limited logistics resources. For operational vaccine delivery planning, we propose using a developed mathematical model that determines the most cost-effective delivery options from supplier to recipients and allows for determining the required number of logistics resources required to fully fulfill the stated volumes. The developed model can also be used as a basis for developing integrated planning models for vaccine delivery from multiple suppliers to different recipients
Keywords: cold chain, delivery method, vaccine logistics, operational planning
JEL-classification: L81, M11, D24
References:
Abramova E.R., Asaliev A.M., Glinskaya M. I. (2024). Sustainability of supply chains in the context of digital business transformation
Ashvin Ashok, Michael Brison, Yann LeTallec (2017). Improving cold chain systems: Challenges and solutions Vaccine. 35 (17). 2217-2223. doi: 10.1016/j.vaccine.2016.08.045.
Azoev G. L., Bykova O.N., Garnov A.P. (2020). Development of economic systems in the digital economy: marketing, services, logistics
Bruce Y. Lee, Leila A. Haidari (2017). The importance of vaccine supply chains to everyone in the vaccine world Vaccine. 35 (35). 4475-4479. doi: 10.1016/j.vaccine.2017.05.096.
De Oliveira L. H., Danovaro-Holliday M. C., Matus C. R., Andrus J. K. (2008). Rotavirus vaccine introduction in the Americas: progress and lessons learned Expert Review of Vaccines. (7). 345-353. doi: 10.1586/14760584.7.3.345.
Haidari L.A., Connor D.L., Wateska A.R., Brown S.T., Mueller L.E. (2013). Augmenting Transport versus Increasing Cold Storage to Improve Vaccine Supply Chains PLOS ONE. 8 (5). e64303. doi: 10.1371/journal.pone.0064303.
Justin R. Ortiz, Joanie Robertson, Jui-Shan Hsu, Stephen L. Yu, Amanda J. Driscoll, Sarah R. Williams, Wilbur H. Chen, Meagan C. Fitzpatrick, Samba Sow, Robin J. Biellik, Kathleen M. Neuzil (2021). The potential effects of deploying SARS-Cov-2 vaccines on cold storage capacity and immunization workload in countries of the WHO African Region Vaccine. 39 (15). 2165-2176. doi: 10.1016/j.vaccine.2021.02.037.
Kartoglu U.H., Moore K.L., Lloyd J.S. (2020). Logistical challenges for potential SARS-CoV-2 vaccine and a call to research institutions, developers and manufacturers Vaccine. 38 (34). 5393-5395. doi: 10.1016/j.vaccine.2020.06.056.
Larin O. N., Matosov M. V. (2024). Vaccine Logistics: Problems and Prospects for Digitalization. Logostics. (10). 46-52.
Lim J., Norman B.A., Rajgopal J. (2022). Redesign of vaccine distribution networks Intl. Trans. in Op. Res.. (29). 200-225. doi: 10.1111/itor.12758.
Lotty Evertje Duijzer, Willem van Jaarsveld, Rommert Dekker (2018). Literature review: The vaccine supply chain European Journal of Operational Research. 268 (1). 174-192. doi: 10.1016/j.ejor.2018.01.015.
Matosov M.V., Larin O.N. (2023). Interaction of participants in the pharmaceutical supply chain. Journal of Economics, Entrepreneurship and Law. 13 (4). 983-994. doi: 10.18334/epp.13.4.117492.
Mercy Mvundura (2017). An economic evaluation of the controlled temperature chain approach for vaccine logistics: evidence from a study conducted during a meningitis A vaccine campaign in Togo The Pan African Medical Journal. 27 (3). 27.
Nader Al Theeb, Mohammed Abu-Aleqa, Ali Diabat (2024). Multi-objective optimization of two-echelon vehicle routing problem: Vaccines distribution as a case study Computers & Industrial Engineering. 187 109590. doi: 10.1016/j.cie.2023.109590.
Nugroho Agung Pambudi, Alfan Sarifudin, Indra Mamad Gandidi, Rahmat Romadhon (2022). Vaccine cold chain management and cold storage technology to address the challenges of vaccination programs Energy Reports. 8 955-972. doi: 10.1016/j.egyr.2021.12.039.
Patrick Lydon, Ticky Raubenheimer, Michelle Arnot-Krüger, Michel Zaffran (2015). Outsourcing vaccine logistics to the private sector: The evidence and lessons learned from the Western Cape Province in South-Africa Vaccine. 33 (29). 3429-3434. doi: 10.1016/j.vaccine.2015.03.042.
Rafael Arevalo-Ascanio, Elena Prozorova, Annelies De Meyer, Roel Gevaers, Ruben Guisson, Wouter Dewulf (2026). Environmental and social assessment of transport and cooling systems in last-mile cold-chain vaccine distribution: A case study in Flanders, Belgium Research in Transportation Economics. 115 101693. doi: 10.1016/j.retrec.2025.101693.
Shi Yu., Lin Yu., Lim M.K., Tseng M.L., Tan Ch., Li Ya. (2022). An intelligent green scheduling system for sustainable cold chain logistics Expert Systems with Applications. 209 118378. doi: 10.1016/j.eswa.2022.118378.
Stef Lemmens, Catherine Decouttere, Nico Vandaele, Mauro Bernuzzi (2016). A review of integrated supply chain network design models: Key issues for vaccine supply chains Chemical Engineering Research and Design. 109 366-384. doi: 10.1016/j.cherd.2016.02.015.
Tan L.J., Barnett M.A., Eisenberg A., Fox Fields H., Hackell J.M., Léger M.M., Meadows B., Moore K.L., Rehm S.J., Rothholz M.C., Smith C., Talkington K., Wexler D.L. (2014). From refrigerator to arm: Issues in vaccination delivery Vaccine. 32 (21). 2389-2393. doi: 10.1016/j.vaccine.2014.02.045.
Terri B. Hyde, Holly Dentz, Susan A. Wang, Helen E. Burchett, Sandra Mounier-Jack, Carsten F. Mantel (2012). The impact of new vaccine introduction on immunization and health systems: A review of the published literature Vaccine. 30 (45). 6347-6358. doi: 10.1016/j.vaccine.2012.08.029.
Thomas D., Motomoke E., Crawford J., Defawe O., Makaya A., Ngwato J.W., Bompongo J., Monzembela J., Ailstock G., Bancroft E., Magadzire B., Baabo D., Watson N. (2021). Optimized supply chain model reduces health system costs in DRC Vaccine. 39 (30). 4166-4172. doi: 10.1016/j.vaccine.2021.05.083.
Tina-Marie Assi, Korngamon Rookkapan, Jayant Rajgopal, Vorasith Sornsrivichai, Shawn T. Brown, Joel S. Welling (2012). How influenza vaccination policy may affect vaccine logistics Vaccine. 30 (30). 4517-4523. doi: 10.1016/j.vaccine.2012.04.041.
Ulla K. Griffiths, Viola S. Korczak, Dereje Ayalew, Asnakew Yigzaw (2009). Incremental system costs of introducing combined DTwP–hepatitis B–Hib vaccine into national immunization services in Ethiopia Vaccine. 27 (9). 1426-1432. doi: 10.1016/j.vaccine.2008.12.037.
Unit 4 : Cold chain and logistics management: Immunization handbook for Medical Officers. Retrieved January 21, 2026, from https://cdn.who.int/media/docs/default-source/searo/india/publications/immunization-handbook-107-198-part2.pdf
World Health Organization et al. Effective vaccine management (EVM) initiative //EVM-setting standards for the Immunization Supply Chain. – 2011. – T. 14.
Xu Sun, Eugenia Ama Andoh, Hao Yu. (2021). A simulation-based analysis for effective distribution of COVID-19 vaccines: A case study in Norway Transportation Research Interdisciplinary Perspectives. 11 100453. doi: 10.1016/j.trip.2021.100453.
Yauba S, Joelle S, Jude N, Tracy BO, Marie K, et al. (2018) Temperature Monitoring in the Vaccine Cold Chain in Cameroon. J Vaccines Vaccin 9: 384. doi:10.4172/2157-7560.1000384, World Health Organization et al. Vaccine management and logistics support //World Health Organization (WHO). – 2020. – S. 1-15
